What is Empirical Formula
The meaning of EMPIRICAL FORMULA is a chemical formula showing the simplest ratio of elements in a compound rather than the total number of atoms in the molecule. COCl2 C O 2 Cl.

Calculating Empirical Formula Chemistry Math Oxygen
This 10-question practice test deals with.

. The empirical formula of a molecule is the smallest whole number ratio of the individual atoms present within that molecule. Here we notice that there are 6 C. Next divide all the masses by their respective.
Then change the to grams. This is because we can divide each number in C 6 H 12 O 6 by 6 to make a simpler whole. We can determine the empirical formula by using the proportion of each element in the compound data.
Luckily the steps to solve either are almost exactly the same. Empirical and Molecular Formula. The molecular formula shows the exact number of different types of atoms.
Solving Empirical Formula Problems There are two common types of empirical formula problems. The empirical formula of a substance can be calculated from the experimentally determined percent composition the percentage of each element present in a pure. The empirical formula of a compound represents the simplest whole-number ratio between the elements that make up the compound.
For example the molecular formula of glucose is C 6 H 12 O 6 but the empirical formula is CH 2 O. In any empirical formula problem you must first find the mass of the elements in the compound. The molar mass of the compound is known to us M 5812 g mol 1.
Empirical formula is known as the formula which contains the simple whole number ratio of each atom that has. An empirical formula is a formula of a compound containing the simplest whole number ratio of atoms present in it. The empirical formula is the first step in the determination of the.
Therefore in chemistry the elements and. An empirical formula represents the simplest whole-number ratio of various atoms present in a compound. The empirical formula of a compound is COCl2 and its molecular mass is 9000u.
A chemical compound n-hexane has a molecular formula of CH3CH2CH2CH2CH2CH3 or simply we can write it as. An Empirical formula is the chemical formula of a compound that gives the proportions ratios of the elements present in the compound but not the actual numbers or. The empirical mass of the compound is obtained by adding the molar mass of individual elements.
How to calculate the Empirical Formula from Element Proportions. In the study of a chemical system we need to represent elements and compounds very frequently. Similarly find out the molecular formula of that compound.
What is the relationship between empirical formula and formula. If given the percent composition of an unknown.

Calculating Empirical Formula Mcat Study Chemistry Labs Calculus
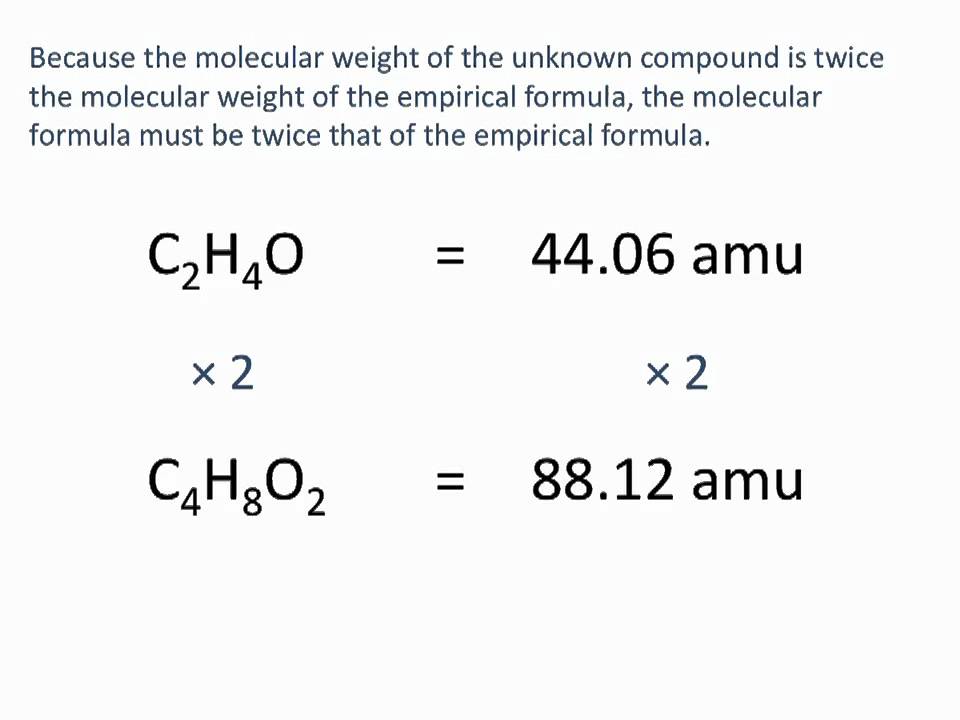
Determining Empirical And Molecular Formulas Chemistry Tutorial Youtube Chemistry Education Chemistry Molecular

How To Calculate An Empirical Formula Chemistry Worksheets Chemistry Notes Scientific Method Worksheet

Determining Empirical And Molecular Formulas Chemistry Tutorial Youtube Chemistry Education Chemistry Molecular
0 Response to "What is Empirical Formula"
Post a Comment